
Two subjects had moderate mitral regurgitation, none of whom developed pulmonary edema. 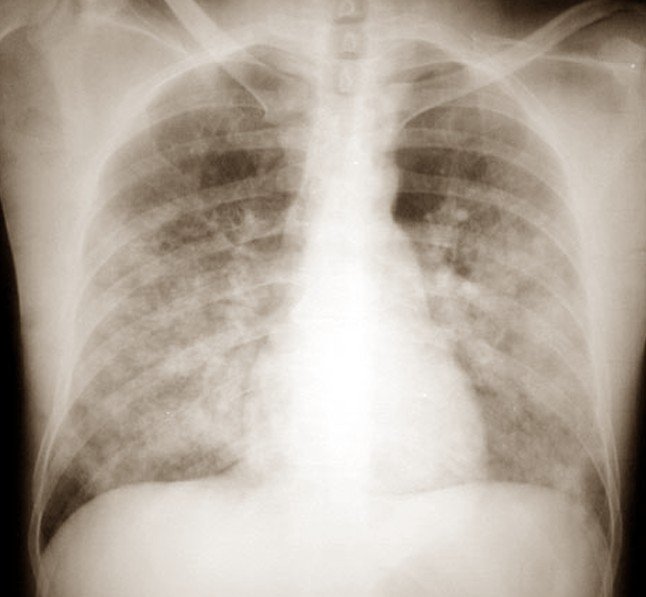
One patient had moderate mitral stenosis with a pulmonary capillary wedge pressure of 11 mm Hg and developed pulmonary edema. The majority of patients (152 of 155) did not have moderate or severe left heart valvular abnormalities on echocardiography. While left ventricular systolic function was normal, approximately one-half of the patients had grade I diastolic dysfunction on echocardiography. The etiology of PAH was mostly idiopathic at 64%. The majority of the study subjects were female (81.9%) with a median age of 51 (39-57) years and white (85%). Table 1 shows the baseline characteristics of the cohort. As per the COMPERA (Comparative, Prospective Registry of Newly Initiated Therapies for Pulmonary Hypertension) study, risk factors for LHD were defined as three or more of the following: history of coronary artery disease, diabetes mellitus, systemic hypertension, BMI ≥ 30, and atrial fibrillation. Factors associated with LHD were extracted in order to determine the cardiovascular profile of the patients. Chest radiography results documented by radiologists were reviewed to ascertain the presence of pulmonary edema. The type of prostacyclin analog, the initiation dates, and doses were extracted and progress notes were meticulously scrutinized to determine the subjective and physical examination findings of pulmonary edema. 10, 11, 12 Chest radiograph signs of pulmonary edema were defined as at least one or more of the following: Kerley B lines, interstitial reticular opacities, vascular cephalization, alveolar infiltrates, or pleural effusions. We defined pulmonary edema as the presence of at least one new symptom (new or worsening dyspnea, orthopnea) or sign (new or worsening rales on pulmonary examination) of pulmonary edema and evidence of new or worsening pulmonary edema on chest radiography. 
Secondary outcomes were duration of ICU and hospital stay, mortality in the hospital and at 3 and 6 months, as well as long-term transplant-free survival. The primary outcome of the study was the development of pulmonary edema within 7 days of initiation of parenteral prostacyclin therapy. These patients were diagnosed and categorized as group 1 PAH as adjudicated by a group of pulmonary hypertension specialists. We retrospectively examined a cohort of adult patients enrolled in the Cleveland Clinic Pulmonary Hypertension Registry (IRB 8097) with a diagnosis of group 1 PAH, and who were initiated on parenteral prostacyclin therapy between 19. We aimed to identify risk factors predisposing toward development of pulmonary edema, and to assess its effect on outcomes including hospital length of stay (LOS) and mortality. We hypothesized that the proportion of patients developing pulmonary edema after initiation of prostacyclin analogs is higher than expected due to LHD and/or unrecognized PVOD/PCH. Besides a pretreatment suspicion of PVOD/PCH, there are no known risk factors for the development of pulmonary edema. It is also uncertain whether the development of pulmonary edema in PAH has any short- or long-term complications. The frequency of pulmonary edema in patients with PAH started on parenteral prostacyclin therapy is not known. 2, 3, 4, 5 However, registries have also shown that a large proportion of patients with PAH have cardiovascular risk factors for left heart disease (LHD), 6 and LHD has been described in patients diagnosed with PAH and a resting pulmonary artery wedge pressure ≤ 15 mm Hg, particularly in systemic sclerosis. It is well recognized that PAH-targeted therapies may unmask pulmonary venoocclusive disease (PVOD) or pulmonary capillary hemangiomatosis (PCH) by causing pulmonary edema. Parenteral prostacyclin therapy is currently recommended for patients with high-risk pulmonary arterial hypertension (PAH), 1 but it can be complicated by the development of pulmonary edema.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
